| (1) |
At absolute zero the structure with the lowest free energy is simply the global potential energy minimum of the PES. At higher temperatures, entropic factors must also be considered. Although we only perform a comprehensive survey of the zero Kelvin geometries, the structural effects of temperature are considered in the subsequent discussion.
To understand the structural effects of the range parameter,
![]() , it is instructive to look more closely at the form of the potential.
The energy can be partitioned into three contributions:
, it is instructive to look more closely at the form of the potential.
The energy can be partitioned into three contributions:
| (2) |
The dominant term in the energy comes from nnn. Ennn is a smaller term and its value varies in a similar manner to nnn. It is only likely to be important in determining the lowest energy structures when other factors are equal. For example, bulk fcc and hexagonal close-packed (hcp) lattices both have twelve nearest neighbours per atom. Next-nearest neighbour interactions are the cause of the lower energy of the hcp crystal when a pair potential such as the LJ form is used.[54,55]
![]() , which measures the energetic penalty for the deviation of a nearest-neighbour distance
from the equilibrium pair distance, is a key quantity in our analysis.
It must not be confused with strain due to an applied external force.
For a given geometry,
, which measures the energetic penalty for the deviation of a nearest-neighbour distance
from the equilibrium pair distance, is a key quantity in our analysis.
It must not be confused with strain due to an applied external force.
For a given geometry, ![]() grows rapidly with increasing
grows rapidly with increasing ![]() because the potential well narrows.
To a first approximation the strain energy grows quadratically with
because the potential well narrows.
To a first approximation the strain energy grows quadratically with ![]() .[38]
Hence, decreasing the range destabilizes strained structures.
.[38]
Hence, decreasing the range destabilizes strained structures.
From the above analysis we can see that minimization of the potential energy involves a balance
between maximizing nnn and minimizing ![]() .
The interior atoms of the three morphologies (Fig. 1) all have twelve nearest neighbours, and
so differences in nnn arise from surface effects.
The optimal shape for each morphology results from the balance between maximizing the
proportion of
.
The interior atoms of the three morphologies (Fig. 1) all have twelve nearest neighbours, and
so differences in nnn arise from surface effects.
The optimal shape for each morphology results from the balance between maximizing the
proportion of ![]() faces (an atom in a
faces (an atom in a ![]() face is 9-coordinate,
but in a
face is 9-coordinate,
but in a ![]() face only 8-coordinate) and minimizing the fraction of atoms in the surface layer.
As Mackay icosahedra (Fig. 1(b)) have only
face only 8-coordinate) and minimizing the fraction of atoms in the surface layer.
As Mackay icosahedra (Fig. 1(b)) have only ![]() faces and are approximately spherical,
the icosahedra have the largest nnn.
Complete Mackay icosahedra occur at
faces and are approximately spherical,
the icosahedra have the largest nnn.
Complete Mackay icosahedra occur at ![]() A pentagonal bipyramid has only
A pentagonal bipyramid has only ![]() faces, but because it is not very spherical
more stable decahedral forms are obtained by truncating the structure parallel
to the five-fold axis to reveal five
faces, but because it is not very spherical
more stable decahedral forms are obtained by truncating the structure parallel
to the five-fold axis to reveal five ![]() faces and then
introducing re-entrant
faces and then
introducing re-entrant ![]() faces between adjacent
faces between adjacent ![]() faces.
The resulting structure is called a Marks decahedron (Fig. 1(c)) and
was first predicted by the use of a modified Wulff construction.[56]
Decahedra generally have lower values of nnn than icosahedra
because of the
faces.
The resulting structure is called a Marks decahedron (Fig. 1(c)) and
was first predicted by the use of a modified Wulff construction.[56]
Decahedra generally have lower values of nnn than icosahedra
because of the ![]() faces.
The tetrahedron and octahedron are fcc structures that have only
faces.
The tetrahedron and octahedron are fcc structures that have only ![]() faces, but
they are not very spherical.
The optimal fcc structure is the truncated octahedron with regular hexagonal faces
(Fig. 1(a)).
At larger sizes than those considered in this study, the optimal structure involves further facetting,
so that it more closely approximates the Wulff polyhedron.[57]
Of the three morphologies fcc structures have the smallest values of nnn.
faces, but
they are not very spherical.
The optimal fcc structure is the truncated octahedron with regular hexagonal faces
(Fig. 1(a)).
At larger sizes than those considered in this study, the optimal structure involves further facetting,
so that it more closely approximates the Wulff polyhedron.[57]
Of the three morphologies fcc structures have the smallest values of nnn.
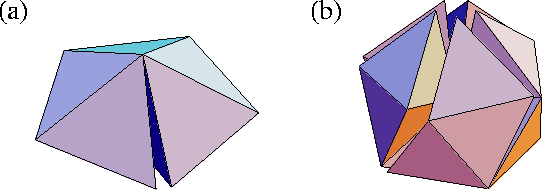 |
A Mackay icosahedron can be decomposed into twenty fcc tetrahedra, but as we see from Fig. 3(b), when twenty regular tetrahedra are packed around a common vertex large gaps remain. To bridge these gaps the tetrahedra have to be distorted, thus introducing strain. The distance between adjacent vertices of the icosahedron is 5% larger than the distance between a vertex and the centre. Similarly, a pentagonal bipyramid can be decomposed into five fcc tetrahedra sharing a common edge. Again, a gap remains if regular tetrahedra are used (Fig. 3(a)) and consequently decahedral structures are strained, although not as much as icosahedra. In contrast, close-packed structures can be unstrained.
Having deduced the relative values of nnn and ![]() for icosahedral, decahedral
and fcc structures, we can predict the effect of the range on the competition between them.
For a moderately long-ranged potential, the strain associated with the icosahedron
can be accommodated without too large an energetic penalty and so such structures are the most stable.
As the range of the potential is decreased, the strain energy associated with icosahedra
increases rapidly, and there comes a point where decahedra become more stable.
Similarly, for a sufficiently short-ranged potential fcc structures become
more stable than decahedral structures.
for icosahedral, decahedral
and fcc structures, we can predict the effect of the range on the competition between them.
For a moderately long-ranged potential, the strain associated with the icosahedron
can be accommodated without too large an energetic penalty and so such structures are the most stable.
As the range of the potential is decreased, the strain energy associated with icosahedra
increases rapidly, and there comes a point where decahedra become more stable.
Similarly, for a sufficiently short-ranged potential fcc structures become
more stable than decahedral structures.
The above decomposition of the potential energy also helps us to understand the effect of size on the
energetic competition between the three morphologies.
The differences in nnn, which arise from the different
surface structures, are approximately proportional to the surface area (![]() ).
The strain energies, however, are proportional to the volume (
).
The strain energies, however, are proportional to the volume (![]() ).
Therefore, the differences in
).
Therefore, the differences in ![]() increase more rapidly with
size than the differences in nnn, thus explaining the change in the most stable
morphology from icosahedral to decahedral to fcc with increasing size.
The effect of increasing the size is similar to the effect of decreasing
the range of the potential: both destabilize strained structures.
increase more rapidly with
size than the differences in nnn, thus explaining the change in the most stable
morphology from icosahedral to decahedral to fcc with increasing size.
The effect of increasing the size is similar to the effect of decreasing
the range of the potential: both destabilize strained structures.