|
Available
Oxford Centre for Soft and Biological Matter
|
Protein and Colloidal Crystallization
The most common method to determine the structure of a protein is by X-ray diffraction of a protein crystal. However, getting proteins to crystallize can be notoriously difficult. Therefore, there is much interest both in understanding why protein crystallization is difficult and in developing methods that can make crystallization easier. Our perspective on the former is that there is an evolutionary selection pressure for proteins in their native state to avoid aggregation or crystallization because these would be potentially harmful to the cell, as is the case for certain types of cataracts and anaemia.One target for colloidal scientists is the development of colloids that can form crystals with open structures, such as diamond, because of the potential photonic applications of these crystals. However, most isotropic colloids form crystals with a high packing fraction. Therefore, there has been much recent interest in generating colloids whose interactions are highly anisotropic. On the other hand, the crystals formed by proteins usually have a low packing fraction, indicating the anisotropic nature of the interprotein interactions.
Our work in this field is focussed in two areas.
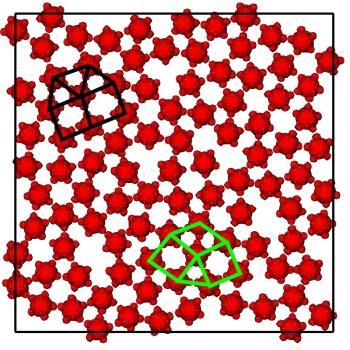 Model systems
Model systems
We are using model "patchy" particles to examine how anisotropy affects crystallization, in particular how the geometry of the patches can be used to aid or hinder crystallization. For example, crystallization is frustrated for particles whose interactions have fivefold symmetry (see right).
The role of lysine
There is a range of evidence that suggests that the amino acid lysine plays an important role in preventing native-state proteins aggregating or crystallizing in vivo. However, the physical mechanism by which lysine achieves this is not yet clear. Lysine is a charged amino acid that is very likely to lie on the surface of the protein, and its long side-chain can have a significant amount of conformational entropy. One suggestion is that the loss of conformational entropy when lysine is involved in a protein-protein interface helps to thermodynamically destabilize the interface. Consistent with this, mutating lysines has been found to often make crystallization eaiser. The aim of this project would be to use atomistic simulations to gain more insight into the mechanisms by which lysine affects interprotein interactions.This project is in collaboration with the group of Dr Ard Louis in The Rudolf Peierls Centre for Theoretical Physics.
Relevant Publications-
E.G. Noya, C. Vega, J.P.K. Doye and A. A. Louis, J. Chem. Phys. 132, 234511 (2010)
The stability of a crystal with diamond structure for patchy particles with tetrahedral symmetry -
E.G. Noya, C. Vega, J.P.K. Doye and A. A. Louis,
J. Chem. Phys. 127, 054501 (2007)
Phase diagram of model anisotropic particles with octahedral symmetry -
J.P.K. Doye, A.A. Louis, I-C. Lin, L.R. Allen, E.G. Noya, A.W. Wilber, H.C. Kok, R. Lyus, Phys. Chem. Chem. Phys. 9, 2197-2205 (2007)
Controlling crystallization and its absence: Proteins, colloids and patchy models -
J.P.K. Doye and W.C.K. Poon, Curr. Opin. Colloid In., 11, 40-46 (2006)
Protein Crystallization in vivo -
J.P.K. Doye, A. A. Louis and M. Vendruscolo, Physical Biology 1, P9-P13 (2004
)
Inhibition of protein crystallization by evolutionary negative design